Abstract
Mining is a devastating operation that not only destroys the natural ecosystem, particularly whether it may be surface mining or underground mining, but also introduces tremendous distortions into the social fabric. The associated problems of waste disposal, water pollution, air pollution, dereliction, vibrations and blasting, land collapse and drop in water table are, of course marked and perhaps to be expected in any mining operation. But little attention has been paid to the fact that mining in remote areas and the impact on health, hygiene nutrition, sanitation, accidents, and increased intensity of work, market prices, prostitution & antisocial activity is a necessary corollary of mining as it is being practiced today”. Present study relates to the impact study of mining on the quality of water which is the main source of drinking water supply to the state capital city that falls in world’s second largest valley- Doon Valley. The present review was focused on impact of phosphorite mining to the water quality of River Bandal of Maldeota. Water samples were collected and were tested in the laboratory for various physico chemical parameters.
License
This is an open access article distributed under the Creative Commons Attribution License which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Type: Research Article
EUR J ENV PUBLIC HLT, Volume 3, Issue 1, 2019, Article No: em0025
https://doi.org/10.29333/ejeph/5839
Publication date: 11 Jul 2019
Article Views: 3705
Article Downloads: 2766
Open Access HTML Content References How to cite this articleHTML Content
INTRODUCTION
Mining is the extraction of valuable minerals or other geological materials from the earth, usually from an ore body, vein of (coal) seam materials recovered by mining includes base metals, precious metals, iron, uranium, coal, diamond, limestone, oil shale, rock salt and potash. Any material that cannot be grown through agricultural processes or created artificially in a laboratory or factory is usually mined. Mining in a wider sense comprises extraction of any non-renewable resource. Mining has a severe impact on environment & ecology of a particular area in both short and long term. It is hazardous enterprise adversely affecting both man and nature. It is one of the main causes of deforestation. In order to mine, trees & vegetation are cleared & burned. The people who are exposed to the toxic waste from the tailings become sick and develop skin rashes headaches, vomiting and diahorrea.
Mining wastes generated from active and inactive mining sites & from beneficiation activities and its impact on human health & the environment are a continuing problem for Govt. entities, private industry & the general public. The nation reported volume of mining waste is immense. The type of mine waste problems are numerous but the most difficult one to address is the Acid mine drainage (AMD) that emanates from both surface & underground mine workings, waste and development rock, tailings piles & ponds. AMD is defined as drainage that occurs as a result of sulphide oxidation in rock exposed to air and water. AMD can be characterized by low pH and in increased acidity elevated heavy metals, sulphate & total dissolved solids (TDS).
Objectives
-
To quantify pollution level in Bandal river from portability point of view.
-
To identify the major sources and types of pollutants in River Bandal.
Study Area
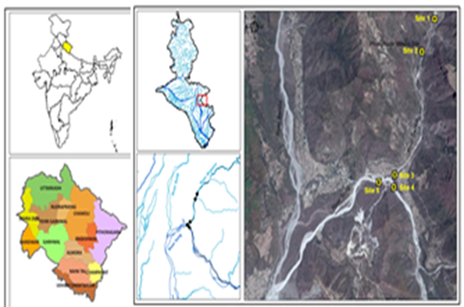
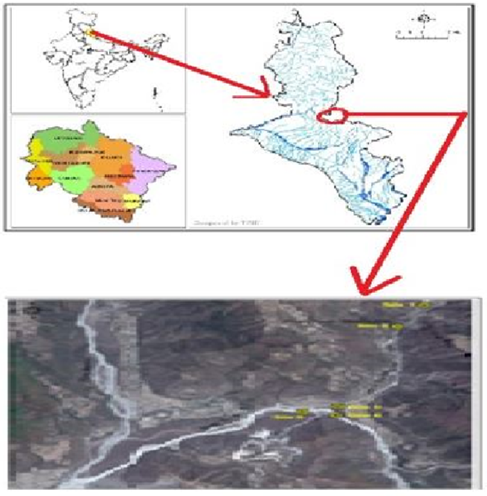
River Bandal Maldeota Dehradun is a perennial river originated from spring fed streams from Surkanda hills of Tehri Garhwal Himalaya. It is one of the most important sources of drinking water for Dehradun city, out of total 30.8 million litre daily availability of water from water sources, the contribution of Bandal River is about 6.75 MLD. Present study has been carried out at Maldeota in Dehradun district, Uttarakhand, India (Figure 2). Maldeota is having an elevation ranging from 650m to about 1050m above mean sea level.
Intensive mining activities in the catchments of Bandal River especially by pyrite phosphorite and Chemical limited (PPCL) near at Maldeota has deteriorated the quality of river water considerably. The present study was undertaken to know the extent of different anthropogenic activities undertaken in the catchments on the river water quality. Hence, about 10 Km stretch of river Bandal before its confluence with river song seems to have severely exposed to mining and mining related activities as well as agricultural runoff cause severe pollution of river water making it unfit for drinking and irrigation uses.
Sampling Sites of Bandal River
The physico-chemical analysis of river Bandal water was conducted from five selected sampling stations before it meet the river Song near at Maldeota. Site I is the Up-stream area where no anthropogenic activities were observed and river water seems to be in natural condition having no anthropogenic pressure and water at this site has been diverted to water Treatment Works of Dehradun meant for drinking water supply in the city. Whereas site II is located just about 5km downstream from the site-I. Site III is Bandal River located just beside the PPCL. Site IV and site V are located before and after confluence of river Bandal and Song. The details of each selected sites are as follows:
|
Table 1. Showing sampling sites of Bandal River
|
Research Site-I (up-stream)
This site is located in the clear water stretch of up-stream about 2 km above the PPCL mining. At this point about 40 per cent of river water has been diverted through canal meant for drinking need of the city. The river also provides water for domestic need such as washing clothes and others for the people residing close to the river. Dense forest vegetation exists in both the banks of river Bandal. The forest of this zone has been demarcated as Reserve Forest by the MOEF. The water at this site appears as crystal clear with its natural colour. Therefore, this site is selected as control site of the study area.
Research Site-II (PPCL effluent drained into Bandal)
This site is about 30 meters below the site 1. Mining operation in surroundings of Koklial Gaon, but here is not limestone but phosphorite that is being mined for Government of India undertaking Pyrite Phosphorite and Chemical Ltd. (PPCL) on the southern flank of the river Bandal. The site was subjected to extensive underground mining previously but now it is officially abandoned after Apex Court Order in 1983.As the mining operation was underground hence mine water still discharged into the river causing considerable contamination to the river water. The effluent was highly turbid and muddy black in appearance.
Research Site-III (Bandal River near at PPCL)
Research Site-III is located at about 200mts downstream from site second. It is located below the bridge on Raipur-Musoorie road. The over burden of solid wastes produced due to mining operation were dumped along the river and the mining water seeping continuously to the river water.
Research Site-IV (Bandal River before confluence of song)
Research Site- IV is located just about 5 km down from the Upstream, near bridge of river Song at Maldeota Village. This site is a confluence point where river Banda! Meets the Song. The catchment area of river Bandal is phosphorite mineral belt. In the past, extensive mining of phosphorite and limestone had been done. However, mining of phosphorite minerals by PPCL had been closed after the Apex Court Order, but illegal mining activities are still going by mining mafia.
Research Site-V (Bandal River after confluence of Song)
This site is located just about half km down from the site IV, near Maldeota Village. This site is a confluence point after river Bandal meets the Song.
Hundreds of trucks load building materials and limestone excavated every day in the area. Furthermore, mining water from abandoned PPCL quarries still discharged into the river Bandal. The survey reveals that this site has been extensively threatened mainly due to mining activities. The forest cover in the catchment of both Song and Bandal seems to have highly degraded with high soil erosion processes in the region.
RESULTS AND DISCUSSION
Based on the monthly data collected for six months (November 2010 to April 2011) from five selected sites of Bandal River, the range and mean values of physico- chemical parameters are depicted in Tables 2-6. Physical test do not directly measure the safety of water supply; however, they do give an indication of its acceptability from portability point of view.
|
Table 2. Variation in Physico- Chemical characteristics of Bandal River water for the month of November 2010 to May 2011
All the values, except pH are in mg/l, otherwise mentioned. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Table 3. Monthly variation in Physico-Chemical characteristics of Bandal River water from Site 1st to 5th
All the values, except pH are in mg/l, otherwise mentioned. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Table 4. Monthly variation in Physico-Chemical characteristics of Bandal River water from Site 1st to 5th
All the values, except pH are in mg/l, otherwise mentioned. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Table 5. Monthly variation in Physico-Chemical characteristics of Bandal River water from Site 1st to 5th
All the values, except pH are in mg/l, otherwise mentioned. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Table 6. Monthly variation in Physico-Chemical characteristics of Bandal River water from Site 1st to 5th
All the values, except pH are in mg/l, otherwise mentioned. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DISCUSSION
Sanitary Condition in Catchments of Bandal River
One of the striking features of stream/river water is the way in which they interact with and are affected by the surrounding environment. Drainage and discharge from the surrounding catchment affect both quantity and quality of water flowing downstream, and it is thus essential that water quality is assessed in the context of catchments condition. From sanitary point of view the condition of sampling sites II, IV and V of Bandal River were observed to be highly unhygienic mainly due to various anthropogenic activities.
General Appearance, Colour and Odour of Bandal Water
The water at upstream was observed natural and transparent while the effluents discharged into the river seems to have caused considerable changes and water appeared muddy-black in appearance. A thin layer appeared on water surface especially in effluent water of PPCL that might be due to presence of pyrite minerals. From the results it is evident that high amount of suspended load as well as floating material brought by surrounding catchments of the mining area may be responsible for impairment of general appearance of water, color and odour.
Transparency
Transparency means water clarity which is opposite of turbidity. It is affected due to presence of suspended matter in water bodies. Presence of high suspended load in water reduce the transparency and may be dangerous from health point of view mainly because there is possibility that harmful substances may be observed. Suspended load clog the gills of fish, reduces their growth rate and decreases the resistance to diseases. According to CPCB guideline Seechi-disc transparency of water bodies (when observed above 100 cm.) should be regarded as excellent” above 76 cm- good, above 50 cm-safe and below 25 cm-unsafe for any use. In the present study Seechi-disc transparency was observed less than 50 cm in mining effluents sites whereas in other sites it was quite good as transparency was above 50 cm.
Temperature
The water temperature fluctuated monthly in the selected sites of Bandal River. The surface water temperature was ranged from 19.0 to 32.0 °C; 17.0 to 32.0 °C; 19.0 to 32.0 °C; 19.6 to 31.0 °C; and 19.0 to 33.0 °C; respectively from site I to Site V. Water temperature closely followed the ambient temperature at all the sites except site II. Bandal is a shallow and narrow river, hence, a rise or fall in the atmospheric temperature due to increase or decrease in solar radiation may be attributed to corresponding rise or fall in atmospheric temperature. The sharp differences between the two may be attributed to presence of dense forest in both the bank of the river that protects heating of river water. Furthermore, the inorganic and organic load present in the water may be responsible for increase in water temperature.
pH
The pH of the river Bandal water in the present study was found to be slightly alkaline to moderate alkaline throughout the study period. It ranged from 6.5 to 7.5; 6.5 to 7.5; 6.0 to 7.0; 6.0 to 7.0; respectively from site I to site V as seen in Tables 2 to 6. The river water seems to have high buffering capacity as evidenced by pH fluctuations within a narrow range. In the present investigation pH values were recorded well within the permissible limit of BIS (1993) set for drinking.
Total Hardness (TH)
The hardness of water is not a pollution indicator parameter but it indicates water quality mainly in terms of calcium and magnesium. Total hardness in the present investigation ranged from 180 to 260 mg/L; 740 to 1100 mg/L; 172 to 200 mg/L; 190 to 482 mg/L; 200 to 460 mg/L; from site I to site V respectively. Total hardness value in the present investigation ranged 460 mg/l to 740 mg/l that crossed the permissible limits of BIS (1991) and WHO (1984) guidelines set for drinking purpose.
Total Alkalinity (TA)
In the present investigation carbonate alkalinity was observed only at site V while on the rest of the sites it was absent. Total alkalinity concentration was ranged from 200.0 to 250 mg/l; 250 to 300 mg/l; 200 to 220 mg/l; 230 to 310 mg/l & 180 to 190 mg/l respectively from site I to site V as shown in Tables 2 to 6.
Chloride (Cl)
In the present study the chloride content in Bandal river ranged in between 20.0 mg/l to 26.0 mg/l and are well within the permissible limit of BIS (1993) standard set for drinking purpose. The mineral Nacl occur widely and are easily dissolved among the halogens, chloride are most energetically stable and vary featly from place to place. The concentration of chloride - an important ion required by the photosynthesizing cells - was fairly low through out the water bodies of Doon Valley.
Phosphate
Agricultural wastes are generally organic materials. It includes crop residues, animal manure, poultry manure, Agricultural wastes also includes the fertilizers and pesticides. The problem of soil erosion in the catchments area of Bandal is acute. However, due to the increased use of commercial fertilizers, as well phosphorite mining effluents containing high phosphate is being discharged into river which greatly influences the biological activities in the riverine system. In the present study the phosphate phosphorus content in Bandal river ranged in between 0.057 to 0.96 mg/l; 0.059 to 0.78 mg/l; 0.56 to 0.67 mg/l; 0.28 to 0.60 mg/l; 0.61 to 0.79 mg/l; respectively from site I to V. The level of phosphate was found well within the permissible limit of BIS (1993) standard set for drinking purpose.
Dissolved Oxygen (DO)
The concentration of dissolved oxygen in an aquatic environment is a function of biological processes such as photosynthesis or respiration and physical process such as water movement and temperature. It does not have any direct bearing on health risks of human and other animals but can predict the health of the water quality of the system. A minimum of 5 mg/L of DO is required to support fish life. In the present study, level of DO never touched optimum level of which is essential for fish growth. It was ranged from 4.9 to 8.10 mg/L in the present investigation. The present study of DO establish a direct impact of organic pollution in the investigated area which indicates that the entire study area completely turned in to nutrient rich condition i.e. heavily eutrophic. The decline of DO (average value of DO mg/l) at PPCL effluent site may be attributed to presence of high organic and inorganic load which deplete the level of DO content at this site.
Total Dissolved Solids
Dissolved solids or Total Dissolved solids denote mainly the various kinds of minerals present in the water. Concentration of dissolved solids is an important parameter in drinking water and other water quality standards. They give a particular taste to the water at higher concentration & also reduce its palatability. High concentration of dissolved solids near 3000 mg/l may also produce distress in cattle and livestock. Plants are also adversely affected by the higher content of solids in irrigation water which increases the salinity of soil.
Conductivity
Conductivity is the measure of capacity of a substance or solution to conduct electric current. Conductivity is reciprocal of resistance. The conductivity of distilled waters ranges between 1 to 5umho/cm but the presence of salts & contamination with waste water increases the conductivity of the water. Consequently a sudden rise in conductivity in the water will indicate addition of some pollutants to it.
In the present study on Bandal river conductivity ranged from 0.171 to 0.550 umho/cm; 0.110 to 0.585 umho/cm; 0.153 to 0.330 umho/cm; 0.272 to 0.340 umho/cm; 0.234 to 0.302 umho/cm; respectively from site I to site V.
CONCLUSION
The overview of the interactions between surface mining and the environment Leads to some fundamental general conclusions. It demonstrates that the Industry’s environmental problems are interconnected and that they have an international character. The solution for environmental problems related to mining involves both the mining company and wider community in action at all levels from personal to international/Governmental. Environmental problems related to mining, particularly the health aspect, soil & water quality are a course of concern taking in consideration the safety of mine workers, surrounding agricultural land and the water quality.
On the basis of present study it may be concluded that although Mining activities have been stopped in Maldeota region by the Apex Court Order, still its effects are seen on the water quality of river Bandal. Hence a comprehensive study should be undertaken so that mining effluent runoff does not contaminate the surrounding areas. The application of ecofriendly mining method and reclamation through proper sustainable manner of this area also reduce the health risks and help in improving the agricultural production and vegetation cover.
References
- APHA (2001). Standard method for the examination of water 17th edition. American public health association, Washington, DC.
- Ashton, P. J., Love, D, Mahachi, H. and Dirks, P. H. (2001). An overview of the impact of min-ing and mineral processing operation on water resources and water quality in the Zambe-zi, Limpopo, and Oilfants catchments in South Africa. Contract report to mining minerals, and sustainable Development project/Southern Africa, CSIREnvironmental; Pretoria, South Africa and University of Zimbabwe, Geology Deptt; Harare, Zimbabwe. Report No. ENV-P-C 2001-042. xvi + 336 pp.
- Bilgrami, K. S., and Dutta Munshi. (1984). Ecology of the river Ganges-Impacts of Human activities and Conservation of Aquatic biota. Proc. 2nd Ann. Workshop of MAB. Projects, New Delhi, 16(17). The Allied Press, Bhagalpur, India.
- BIS (1983). Bureau of Indian standard for drinking water. IS: I0500-1993, New Delhi.
- Brown, M. T. and Odum, H. T. (1985). Studies of a method of wetland reconstruction following phosphate mining. Final report (No. PB-87-173175/XAB). Florida Univ., Gainesville (USA). Center for Wetlands.
- Calmels, D., Gaillardet, J., Brenot, A. and France Lanord, C. (2007). Sustained Sulfide Oxidation by physical erosion processes in the Mackcnzie River basin. Climatic Perspectives, Geology, 35(11), 1003-1006. https://doi.org/10.1130/G24132A.1
- Cappuyns, V., Swennen, R. Vandamme, A. and Niclaes, M. (2006). Environment Impact of the formal Pb-zn mining and smelting in east Belgium. Journal of geochemical exploration, 88(1-3), 6–9. https://doi.org/10.1016/j.gexplo.2005.08.005
- Colmer, A. R. and Hinkle, M. E. (1947). The role of micro-organisms in acid mine drainage-a preliminary Report. Science, 106(2751). https://doi.org/10.1126/science.106.2751.253
- Das Gupta, S, Tiwari, B. K. and Tripathi, R. S. (2002). Coal Mining in Jainthia Hills, Meghalaya: An Ecological perspective. In; Jainthia Hills, A Meghalaya Tribe: Its Env., land and people (Eds. P.M. Passah and A.S. Sarma) (pp.121-128). New Delhi: Reliance Publishing House.
- Hynes, H. B. N. and Pentelow, F. T. K. (1960). The biology of polluted waters (Vol. 202). Liverpool: Liverpool university press.
- Jha, A. K. and Singh, J. S. (1991). Spoil characteristics and vegetation development of an age series of mine spoils in a dry tropical environment. Vegetatio, 97(1), 63–76. https://doi.org/10.1007/BF00033902
- Johnson, S. W., Drice, S. and Adam Moles, D. (1997). Effects of submarine Mine Tailings Dis-posal on Juvenile yellow fin sole (Pleuronectas aspen): A laboratory study. Marine Pollution Bulletin, 36(4), 278-287. https://doi.org/10.1016/S0025-326X(97)00181-1
- Kumar, R., 1996, May. Impact of silica sand mining activity on environment in Faridabad district, Haryana. In Symposium on earth sciences in environmental assessment and management, Bangalore, India (Vol. 17).
- Lerman, A. and Wu, L. (2006). Co2 and sulfuric acid Controls of Weathering and river water composition. Journal of Geochemical Exploration, 88(1-3), 427-430. https://doi.org/10.1016/j.gexplo.2005.08.100
- Lerman, A., Wu, L. L, and Mackenzie, F. T. (2007). CO2 and H2SO4 Consumption in Weathering and material transport to the ocean, and their role in the global carbon balance. Marine Chemicals, 106(1-2), 326-350. https://doi.org/10.1016/j.marchem.2006.04.004
- Leveling, B. R. and Wylie, R. W. (1993). Water Resources Investigation. Report 93-4002, VA.
- Mason, R. P. (1997). Mining Waste Impacts on Stream Ecology, In C. D. Da Rosa (ed). Golden Dreams, Poisoned Streams, How Reckless Mining Pollutes America’s Waters & How We Can Stop It. Washington D.C, Mineral Policy Center. (ISBN 1-889617-01-6).
- Mathews, T. D. (1980). Ecological characterization of the Sea Island coastal region of South Carolina and Georgia. Vol. I, physical features of the characterization area (No. 79/40). US Fish and Wildlife Service.
- Mortiner, C. H. (1941). The exchange of dissolved substances between mud and water in lakes. Journal of Ecology, 29(2), 280-329. https://doi.org/10.2307/2256395
- Pulles, W., Health, R. and Howard. M., (1996). A manual to access and manage the impact of Gold Mining Operations on the Surface Water. Environment Water Research Commision Report. No.TT 79/96, Pretoria, South Africa.
- Ratha, D. S. and Venkataraman, G. (1997). Application of statistical methods to study Seasonal variation in the mine contaminants in soil and ground water, Goa, India. Environmental Geology, 29(1-3), 253-262. https://doi.org/10.1007/s002540050124
- Ripley, E. A. (1996). Environmental Effects of Mining. Delray Beach, Florida: St. Lucie Press.
- Trivedy, R. K. and Goyal, P. K. (2002). Examination of Chemical and Biological Methods for Water Pollution Studies. Environmental Publications in Karad, India, 211-215.
How to cite this article
Vancouver
Bhat BB. Impact of Phospharite Mining of River Bandal Dehradun. EUR J ENV PUBLIC HLT. 2019;3(1):em0025. https://doi.org/10.29333/ejeph/5839
APA
Bhat, B. B. (2019). Impact of Phospharite Mining of River Bandal Dehradun. European Journal of Environment and Public Health, 3(1), em0025. https://doi.org/10.29333/ejeph/5839
AMA
Bhat BB. Impact of Phospharite Mining of River Bandal Dehradun. EUR J ENV PUBLIC HLT. 2019;3(1), em0025. https://doi.org/10.29333/ejeph/5839
Chicago
Bhat, Bilal Bashir. "Impact of Phospharite Mining of River Bandal Dehradun". European Journal of Environment and Public Health 2019 3 no. 1 (2019): em0025. https://doi.org/10.29333/ejeph/5839
Harvard
Bhat, B. B. (2019). Impact of Phospharite Mining of River Bandal Dehradun. European Journal of Environment and Public Health, 3(1), em0025. https://doi.org/10.29333/ejeph/5839
MLA
Bhat, Bilal Bashir "Impact of Phospharite Mining of River Bandal Dehradun". European Journal of Environment and Public Health, vol. 3, no. 1, 2019, em0025. https://doi.org/10.29333/ejeph/5839

 Full Text (PDF)
Full Text (PDF)